10. One of the metals below will react violently when put in water. Which one?
- A. Aluminium
- B. Copper
- C. Potassium
- D. Iron
|
|
11. Which of these lists has metals in the correct order from most reactive to least reactive?
- A. sodium, zinc, copper
- B. zinc, copper, sodium
- C. iron, sodium, copper
- D. sodium, copper, zinc
|
|
12-15. When iron pieces are added to copper sulfate, a reaction occurs and copper metal is left at the bottom of the tube: |
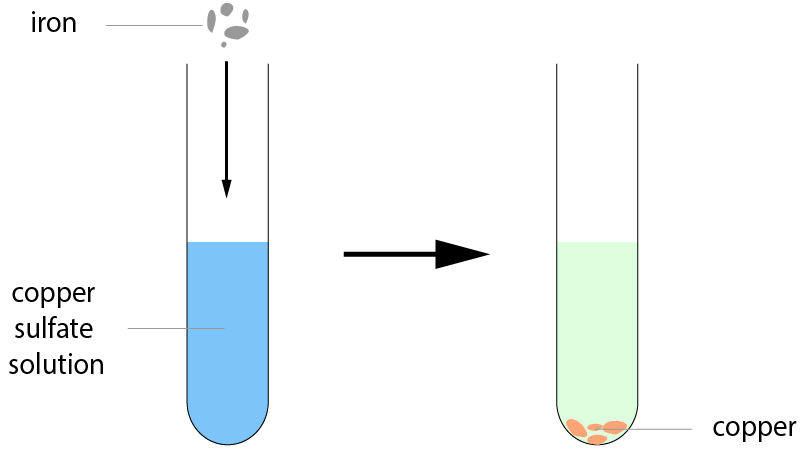 |
12. Which sentence best describes why this happens?
- A. Iron is more reactive than sulphur.
- B. Iron is more reactive than copper.
- C. Copper is more reactive than sulphur.
- D. Copper is more reactive than iron.
|
|
13. What is the name given to this kind of reaction?
- A. Neutralisation reaction.
- B. Displacement reacton.
- C. Combustion.
- D. Thermal decomposition.
|
|
14. Which of these word equations correctly shows this reaction?
- A. iron + copper sulfate → copper + hydrogen + water
- B. iron + copper sulfate → copper + a salt + water
- C. iron + copper sulfate → copper + iron + sulfur
- D. iron + copper sulfate → copper + iron sulfate
|
|
15. The test tube in this experiment gets hot - the chemical reaction produces heat energy. What is this kind of reaction called?
- A. endothermic
- B. exothermic
- C. thermal decomposition
- D. neutralisation
|
|
16. If carbon is added to a metal oxide and heated, then it can displace the metal producing carbon dioxide and a pure metal.
However, only one of the reactions below can occur . Which one?
- A. carbon + sodium oxide → sodium + carbon dioxide
- B. carbon + magnesium oxide → magnesium + carbon dioxide
- C. carbon + aluminium oxide → aluminium + carbon dioxide
- D. carbon + iron oxide → iron + carbon dioxide
|
|
|
