| 11. Changes can be either physical or chemical.
Which of the following changes is physical and which is chemical?
| |
Physical change |
Chemical change |
| A |
Melting wax |
Decomposing chalk |
| B |
Melting wax |
Condensing steam |
| C |
Burning methane |
Decomposing chalk |
| D |
Burning methane |
Condensing steam |
|
|
| 12. P, Q, R, S and T are five food colourings. The dyes in the food colourings were separated by paper chromatography and the results are shown below:
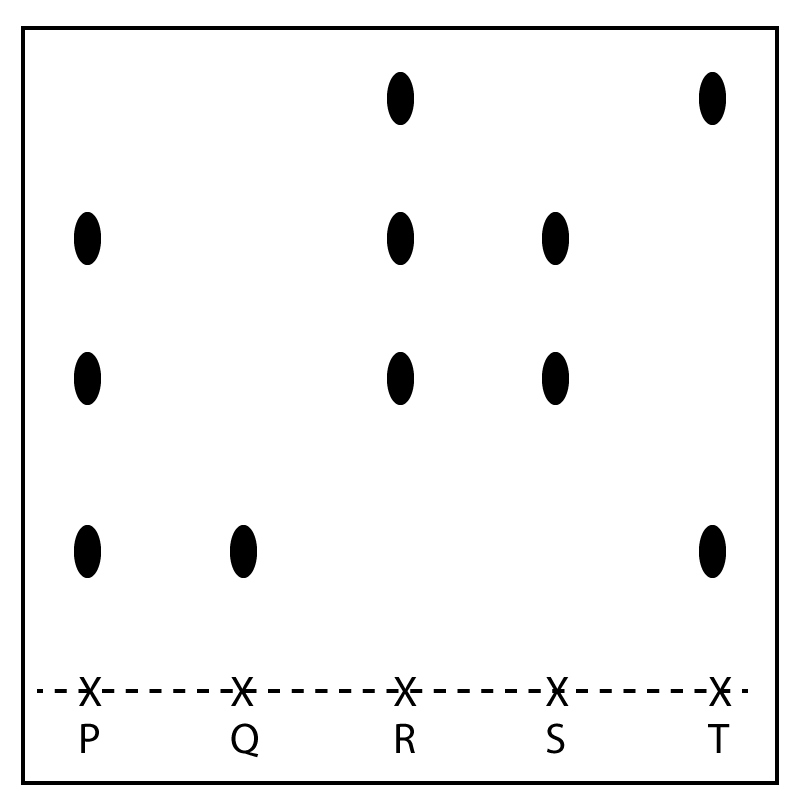
|
|
Which food colourings could be mixed to produce food colouring P?
- A. Q and R
- B. Q and S
- C. Q and T
- D. R and T
|
|
13. Which of the following statements about mixtures is true?
- A. A mixture can only be separated by chemical reactions
- B. The chemical properties of each substance in a mixture are altered on mixing
- C. A mixture consists of two or more substances not chemically combined
- D. A mixture can be represented by a formula
|
|
14. Which method could be used to separate a mixture of salt and water to obtain BOTH parts of the mixture?
- A. filtration
- B. evaporation
- C. distillation
- D. crystallization
|
|
Q15-16:
A student was investigating the dyes present in a food colouring using chromatography. He set up his chromatogram as shown in the diagram:
|
 |
15. Where should the student place the food colouring?
- A. At the top of the paper
- B. on the line
- C. Below the line
- D. In the solvent
|
|
16. Which statement about this experiment is correct?
- A. The line should be drawn in ink
- B. The solvent should be above the line
- C. The dyes must differ in their boiling point
- D. The dyes must differ in their solubilities in the solvent
|
|
| 17. In chromatography, how many spots are produced by a pure substance?
|
|
Q18-20:
Several different methods are used to separate mixtures.
Select the best method to separate .. |
|
18. Drinking water from sea water.
- A. filtration
- B. distillation
- C. crystallization
- D. chromatography
|
|
19. excess solid from the mixture formed by reacting excess solid copper(II)carbonate and dilute hydrochloric acid.
- A. filtration
- B. fractional distillation
- C. crystallization
- D. chromatography
|
|
20. solid copper(II)chloride from a copper(II)chloride solution.
- A. filtration
- B. fractional distillation
- C. crystallization
- D. chromatography
|
|
|
